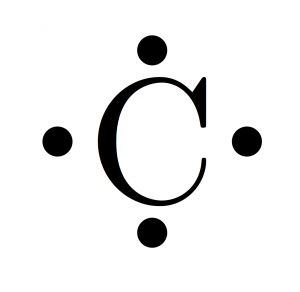
Another way to find an element's valence electrons is with something called an electron configuration. Learn how to read an electron configuration. This means that an atom can have multiple numbers of valence electrons depending on how it is manipulated. For reasons that are a little too complex to explain here, when electrons are added to the outermost d shell of a transition metal (more on this below), the first electrons that go into the shell tend to act like normal valence electrons, but after that, they don't, and electrons from other orbital layers sometimes act as valence electrons instead. 
Generally, the valence electrons are the electrons in the outermost shell - in other words, the last electrons added. As electrons are added to an atom, they are sorted into different "orbitals" - basically different areas around the nucleus that the electrons congregate in.See below for a quick run-through or skip this step to get right to the answers. Understanding why transition metals don't really "work" like the rest of the periodic table requires a little explanation of the way electrons behave in atoms. Get the free view of Chapter 5, Inside the Atom Science 8th Standard Maharashtra State Board additional questions for Mathematics Science 8th Standard Maharashtra State Board Maharashtra State Board,Īnd you can use to keep it handy for your exam preparation.Understand that transition metals don't have "traditional" valence electrons. Maximum Maharashtra State Board Science 8th Standard Maharashtra State Board students prefer Balbharati Textbook Solutions to score more in exams. The questions involved in Balbharati Solutions are essential questions that can be asked in the final exam. Using Balbharati Science 8th Standard Maharashtra State Board solutions Inside the Atom exercise by students is an easy way to prepare for the exams, as they involve solutionsĪrranged chapter-wise and also page-wise. Thomson’s Atomic Model, Lord Rutherford’s Atomic model, Neils Bohr’s Model of an Atom, History of Atom, Dalton’s atomic theory, Atoms: Building Blocks of Matter, Discovery of Charged Particles in Matter, Nucleus, Electrons (e), Atomic Mass, Atomic Number (Z), Mass Number (A), and Number of Neutrons (n), Electronic Configuration of Atom, Valency, Isotopes, Nuclear Reactor. Balbharati textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.Ĭoncepts covered in Science 8th Standard Maharashtra State Board chapter 5 Inside the Atom are Structure of an Atom, Protons (p), Neutrons (n), J. 
This will clear students' doubts about questions and improve their application skills while preparing for board exams.įurther, we at provide such solutions so students can prepare for written exams. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion.īalbharati solutions for Mathematics Science 8th Standard Maharashtra State Board Maharashtra State Board 5 (Inside the Atom) include all questions with answers and detailed explanations. has the Maharashtra State Board Mathematics Science 8th Standard Maharashtra State Board Maharashtra State Board solutions in a manner that help students
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
